In industry, the rare-earth elements (REEs) comprise the 14 naturally occurring lanthanide elements, plus yttrium. These elements are generally unfamiliar but are essential for a large number of applications that affect the daily lives of most everyone (see table). Up until the early 1980s, the United States was the dominant global producer of REE ores and derived products but has since been displaced by China, even as global REE production has more than doubled. Recent restrictions on REE exports from China have raised concern over the reliability of China as a supplier of REE raw materials and stimulated exploration and new mine development activity for REEs outside of China.
Atomic symbol |
Element |
Percent share in use* |
Principal uses in 2008 |
|---|---|---|---|
La |
Lanthanum |
32.7 |
Fluid catalytic cracking (46%), battery alloys (16%), glass polishing (13%), metallurgy (8%), glass additives (7%), ceramics (3%), phosphors (2%), automobile catalytic converters (1%), fuel cells |
Ce |
Cerium |
29.9 |
Glass polishing (25%), glass additives (19%), automobile catalytic converters (16%), metallurgy (14%), battery alloys (10%), fluid catalytic cracking (5%), phosphors (2%), ceramics (2%) |
Pr |
Praseodymium |
6.8 |
Magnets (70%), metallurgy (7%), glass polishing (7%), ceramics (5%), battery alloys (5%), automobile catalytic converters (2%), glass additives (1%) |
Nd |
Neodymium |
18.5 |
Magnets (76%), metallurgy (8%), battery alloys (5%), ceramics (4%), glass additives (2%), automobile catalytic converters (1%), superconductors |
Sm |
Samarium |
0.4 |
Battery alloys (73%), magnets, catalysts, glass additives, ceramics, fuel cells |
Eu |
Europium |
0.3 |
Phosphors (100%) |
Gd |
Gadolinium |
1.0 |
Magnets (69%), phosphors (21%), metallurgy, nuclear reactor shielding |
Tb |
Terbium |
0.4 |
Phosphors (89%), magnets (11%), fuel cells, magnetic sensors |
Dy |
Dysprosium |
1.0 |
Magnets (100%) |
Ho |
Holmium |
† |
Glass additives, magnetic flux concentrators, nuclear fuel control rods |
Er |
Erbium |
† |
Glass additives, phosphors, metallurgy |
Tm |
Terbium |
† |
Fuel cells, phosphors, magnetic sensors |
Yb |
Ytterbium |
† |
Glass additive, gamma ray source, metallurgy, stress gauges |
Lu |
Lutetium |
† |
Phosphors, magnetic bubble memory devices, positron emission tomography detectors, medical research |
Y |
Yttrium |
9.0 |
Phosphors (54%), ceramics (32%), glass additives (2%) |
*As rare-earth oxide (REO), percent share in total REO use.
†These elements combined comprise 0.4% of REO consumption.
Source:Data from T. G. Goonan (2011).
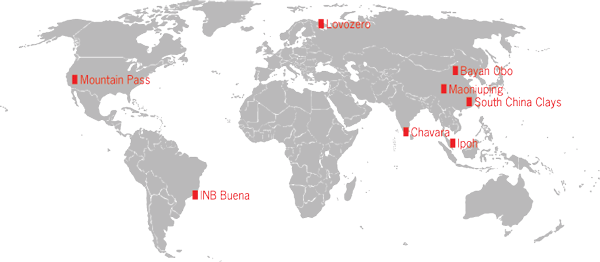
The REEs were first commercialized during the 1880s, when rare-earth oxide mixtures were used to make incandescent mantles for gas lights. The REEs were extracted from monazite mined from coastal heavy-mineral placer deposits in Brazil and India. Similar mixtures found applications in steel making. The discovery of the Mountain Pass REE deposit in California in 1949, one of the largest REE deposits in the world, provided a potential source of supply much larger than was required by industry at the time (Fig. 1). Molycorp, owner of the property, developed a small mine and undertook significant research to find commercial applications for REEs. Among the new uses that emerged at the time were REE catalysts for refining crude oil, cerium oxide for polishing glass, and REE phosphors for color televisions. To meet this emerging demand, Molycorp greatly expanded production during the early 1960s, establishing itself as the dominant producer. Meanwhile, the world's largest REE deposit had been found in China at Bayan Obo in Inner Mongolia, which became the basis for China's emergence as the current dominant producer of REEs, reaching over 90% of world production by 2000 (Fig. 2).
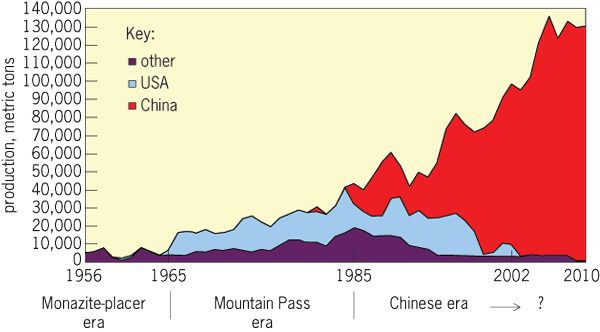
New applications for REEs continued to emerge, including REE magnets, ceramic capacitors, optical glass, and nickel-hydride batteries. REEs are generally expected to play an important role in many alternative energy technologies, and demand for REEs is growing rapidly. By some estimates, the overall rate of demand growth is about 8% per year, which is an unusually high rate for a mineral commodity. Meanwhile, China, the dominant producer, has imposed successively smaller quotas on exports of REE raw materials and some intermediate products, placing severe restrictions on supply. Prices of REE materials outside China have risen sharply, causing many consumers to seek alternatives. There is a general consensus among policy makers in the advanced industrial economies of Asia, Europe, and North America that alternative sources of REE supply need to be developed outside China.
REE mineral deposits
The REEs have very similar chemical properties and thus occur together in nature. Within the Earth's crust, the abundance of each REE generally decreases as its atomic number increases. The REEs are conventionally subdivided into the light REEs (lanthanum through europium) and the heavy REEs (gadolinium through lutetium plus yttrium). Where geologic processes have concentrated REEs into deposits that are economic to mine, light REE-rich deposits are the most common and deposits that are relatively enriched in heavy REEs are rare. Overall, the crustal abundance of the REEs, estimates of which range from 150 to 220 ppm, exceeds that of many other metals that are mined on a large scale, such as copper (55 ppm) and zinc (70 ppm). Despite this favorable crustal abundance, REE deposits are quite uncommon and usually modest in size.
The principal source of REEs are deposits hosted by carbonatites, unusual intrusive rocks composed of at least 50% carbonate minerals. Carbonatites are found emplaced within continental rift zones where a thinned and structurally fractured crust allows melted mantle to reach the surface. At Mountain Pass, California, the REE carbonate mineral bastnaesite (bastnäsite) is an essential rock-forming mineral and the carbonatite contains 8% total REEs. Most known carbonatites contain significantly less REEs, generally at concentrations too low for economic extraction. At Mount Weld, Western Australia, deep weathering has concentrated REEs in parts of the weathered zone to a level similar to that at Mountain Pass. The Bayan Obo deposit in China, the nature and origin of which is still debated, appears to be an overprinting of carbonatite-REE mineralization on parts of an iron deposit.
A broader class of alkalic igneous rocks, sometimes associated with carbonatites, also hosts REE mineralization that has sometimes been mined. These deposits are generally lower in REE grade and are often mineralogically complex, which presents problems for economic extraction. Many of these deposits are quite large and are under active exploration and metallurgical testing. REE-rich monazite is found in many coastal heavy-mineral deposits that are now mined for titanium and zircon, but the monazite is usually enriched in uranium and thorium and few producers are willing to take on the costs of processing the radioactive materials. Deep weathering of some granites in south China has yielded lateritic soil with about 0.3% REEs, often relatively enriched in heavy REEs. Despite the low grade, the clayey material is cheaply extracted and need only be leached to remove the REEs. Exploration for similar deposits outside China is currently underway.
Several key factors determine the economic viability of a REE deposit. Distance from existing infrastructure, especially the electrical grid, significantly affects capital and operating costs. A very remote mine will have to generate the significant power required for processing REE-bearing material, onsite, with diesel-fired plants. A deposit located at or near the surface can be mined by open-pit methods at considerably less expense than the underground methods required for deeply buried deposits. Grade, the proportion of the material to be mined that contains rare earths, determines how much material must be processed to obtain a given amount of REE product, a key factor in economic efficiency. Size and type of REE minerals is also critical. The smaller the mineral size, the more finely the rock must be crushed and ground to liberate the REE-bearing mineral, at significantly higher energy cost. Some REE minerals are readily leached with relatively inexpensive, commercially available chemicals, while others have no commercially tested treatment processes available.
Mining and mineral processing
REE-bearing rock must first be removed from the ground and the REE content concentrated before chemical methods of REE extraction and separation may be applied. Most rare-earth ores are mined by conventional open-pit methods in which rock is broken by blasting, loaded onto trucks with large shovels, and hauled to a concentration plant. Concentrating is by physical separation of the REE-bearing minerals from all other minerals in the rock. The ore is crushed and ground in multiple stages until most of the rare-earth minerals interlocked with the other minerals are broken free. Next, in a method known as froth flotation, the rare-earth minerals are coated with a chemical that repels water and allows them to float to the surface attached to air bubbles in agitated tanks, where they are skimmed off as a concentrate. The remaining minerals are disposed of as waste and the REE concentrate is treated onsite or sent to another location for extraction and separation. Research is underway to improve the flotation process, perhaps with better reagents, to allow the economic treatment of lower-grade REE ores.
Separation and commercialization
Although there is a commercially available mixed REE oxide product known as mischmetal, most REEs are recovered as individual rare-earth oxides (REOs) and marketed as such or used to make other REE chemicals or metals and alloys. The REE concentrate is leached with an acid and the resulting REE-rich solution processed through sequential steps to recover individual REEs. Cerium can be recovered by addition of sodium hydroxide, which causes the cerium to drop out of solution as an oxide or hydroxide. The other REEs are typically separated by solvent extraction, a process in which an organic chemical specially designed to extract a particular REE is forced countercurrent to the REE-bearing leach solution. The REE desired passes into the organic phase, which is separated from the leach solution, and the REO is recovered by stripping with acid. Multiple solvent extraction steps are required because of low separation efficiencies.
No individual REE plant recovers every REE present in the ore. Many of the REEs present, particularly the heavy REEs, are too low in concentration for economic recovery. Some of the heavy REEs, such as lutetium, have very small markets and are unlikely to be recovered even from one of the rare deposits that are relatively rich in heavy REEs. Research is underway to improve the separation of REEs, particularly through the development of more efficient extractants. Many of the advanced methods developed for recovering actinide elements from radioactive wastes, such as supercritical fluid extraction, should be applicable to the lanthanide elements. However, work in this area has only just started.
The reduction of REOs to rare-earth metals is very difficult because of the high stability of REO compounds. The present method, used at only a few plants around the world, is a highly complex refining method using chlorides as a reductant. Molten-salt electrolysis and other electrorefining methods are under investigation as more economic alternatives.
Environmental challenges
Mining and concentration of REE ores presents conventional problems of waste rock and concentrator waste disposal, which are generally handled through careful engineering of onsite waste-disposal facilities. When mining ceases, all equipment and processing facilities are removed, and waste-disposal facilities are recontoured, capped with topsoil, and revegetated. Water flow onsite is directed to prevent interaction with waste materials. Open pits are typically left unfilled. The rock that was blasted and removed expands by about 40% by volume and thus will not all fit back in. If backfilled with waste rock, any inflowing ground and surface waters will encounter a highly porous and permeable fill, which greatly increases water–rock interaction and, depending on the rock chemistry, has the potential for significant water contamination. In nonarid climates, a pit lake may form and may develop undesirable water chemistry through water–rock interactions, requiring ongoing water treatment. These best practices are widely used outside China, which has only recently begun to address environmental problems in its REE industry.
A modern REE separation plant, such as that constructed at Mountain Pass, California, in 2012, recycles and regenerates chemicals and water used and even produces power from the waste heat generated from the exothermic chemical reactions involved. Any waste generated is rather small and is disposed of onsite with proper sealing to prevent ground- or surface-water contamination. No smelting is involved, so air-pollution issues are limited to dust generation during mining and from any onsite power-generation facilities. The final REE products are shipped in sealed containers of various kinds.
Outlook for mine development
As of 2012, two REE mines were under development outside China, and about a dozen other REE deposits were at various stages of advanced exploration and economic feasibility studies. The two new mines are Mount Weld in Australia and Mountain Pass in California, the latter being a redevelopment of an older, inactive mine (Fig. 3). Mount Weld ships its REE concentrates to a plant in Malaysia for extraction and separation. Mountain Pass extracts its REEs onsite and produces a wide range of REE chemical products. As for the other projects, whether and when they will become producing mines depends on the economic viability of the deposit itself, the time and expense required for environmental permitting, how much demand increases and Chinese exports decrease in the future, as well as how quickly competitors get their new mines into production.
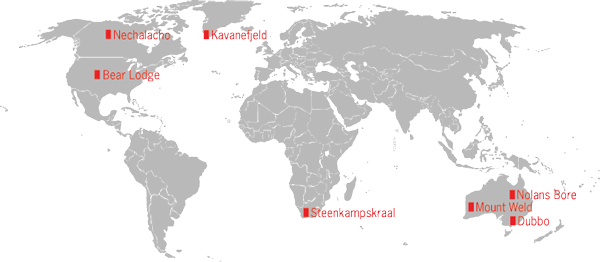
A number of these other projects have recently released positive economic feasibility studies. This is a necessary, but not sufficient, condition for investment in a new mine. The various sources of financing of new mines can compare projects as well as assess the need for new capacity and will invest where returns are commensurate with risks. A review of the various possibilities by this author found that supplies of REEs will likely remain tight through 2015, if not longer, simply because of the time required to permit and develop new mines. In the long run, there do appear to be substantial REE resources of sufficient quality to compensate for the lost Chinese exports and to meet increasing demand. The challenge for future producers is to keep costs competitive with China's and perhaps invest downstream to obtain captive markets for REE mine products.
See also: Carbonatite; Electrolysis; Electrometallurgy; Heavy minerals; Lanthanide contraction; Laterite; Monazite; Open-pit mining; Rare-earth elements; Rare-earth minerals; Yttrium





