A new single-dose antiviral drug can shorten the duration of the flu in teens and adults by about a day, according to a study published in the New England Journal of Medicine (September 2018). The drug, baloxavir marboxil (Xofluza™; baloxavir), developed by the Japanese pharmaceutical company Shionogi, shortened flu symptoms similarly to that of oseltamivir (Tamiflu®), which is normally taken twice daily for five days. Compared to oseltamivir, baloxavir showed a greater reduction in the viral load one day after starting treatment. No significant side effects were reported for baloxavir. In addition, baloxavir was found to be effective against both influenza A and B viruses, including strains resistant to antiviral drugs. See also: Influenza; Virus
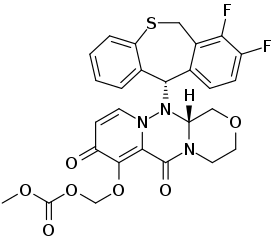
Baloxavir inhibits the enzyme known as influenza polymerase, preventing the influenza virus from replicating. Influenza antiviral drugs, such as oseltamivir and zanamivir (Relenza®), inhibit the neuraminidase enzyme, preventing the entry and release of influenza viruses from cells. See also: Drug resistance; Enzyme; Enzyme inhibition
In Japan, baloxavir was approved for use in children and adults in February 2018. The U.S. Food and Drug Administration (FDA) began reviewing baloxavir in June 2018 and approved it for use on October 24, 2018. However, to avoid contracting the flu, the U.S. Centers for Disease Control and Prevention (CDC) recommend annual influenza vaccination for everyone age 6 months and older. See also: Public health; Vaccination





